Client Case Study

FDA Support
Verification
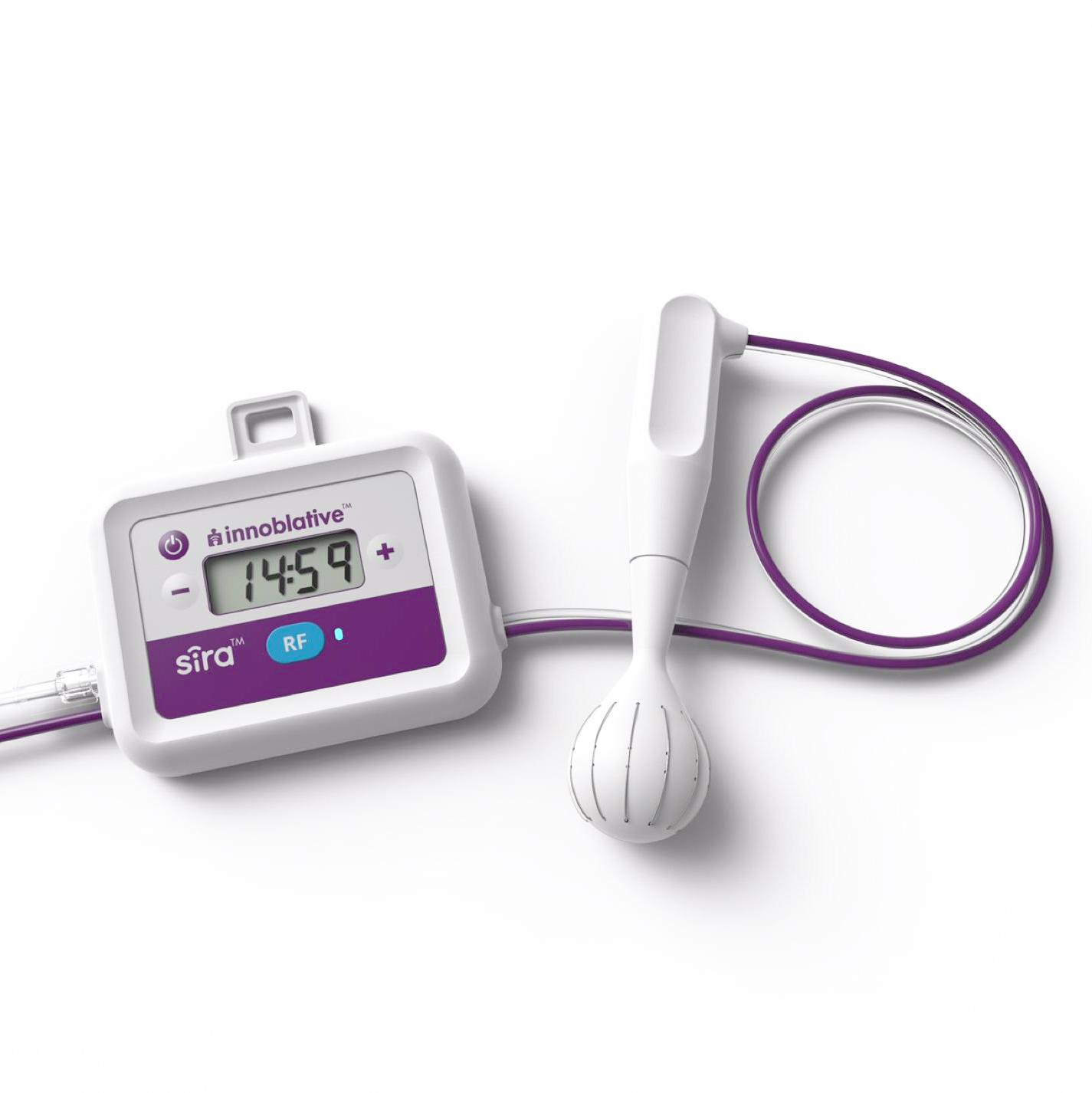
Innoblative received Breakthrough Device Designation from the FDA for the company's SIRA™ RFA Electrosurgical Device (SIRA). The SIRA device is intended for use in breast cancer patients undergoing BCS, commonly referred to as lumpectomy. Innoblative's SIRA™ device has a unique spherical shape to the entire cavity that circumferentially delivers RF energy and yields reproducible ablation depths to provide greater confidence of a consistent thermal effect.
What we delivered:
- Design verification support & test protocols
- Design Root Cause Analysis
- Hands-on clinical device testing support
- 510(k) application documentation support







.svg)
