Client Case Study

Development
FDA Support
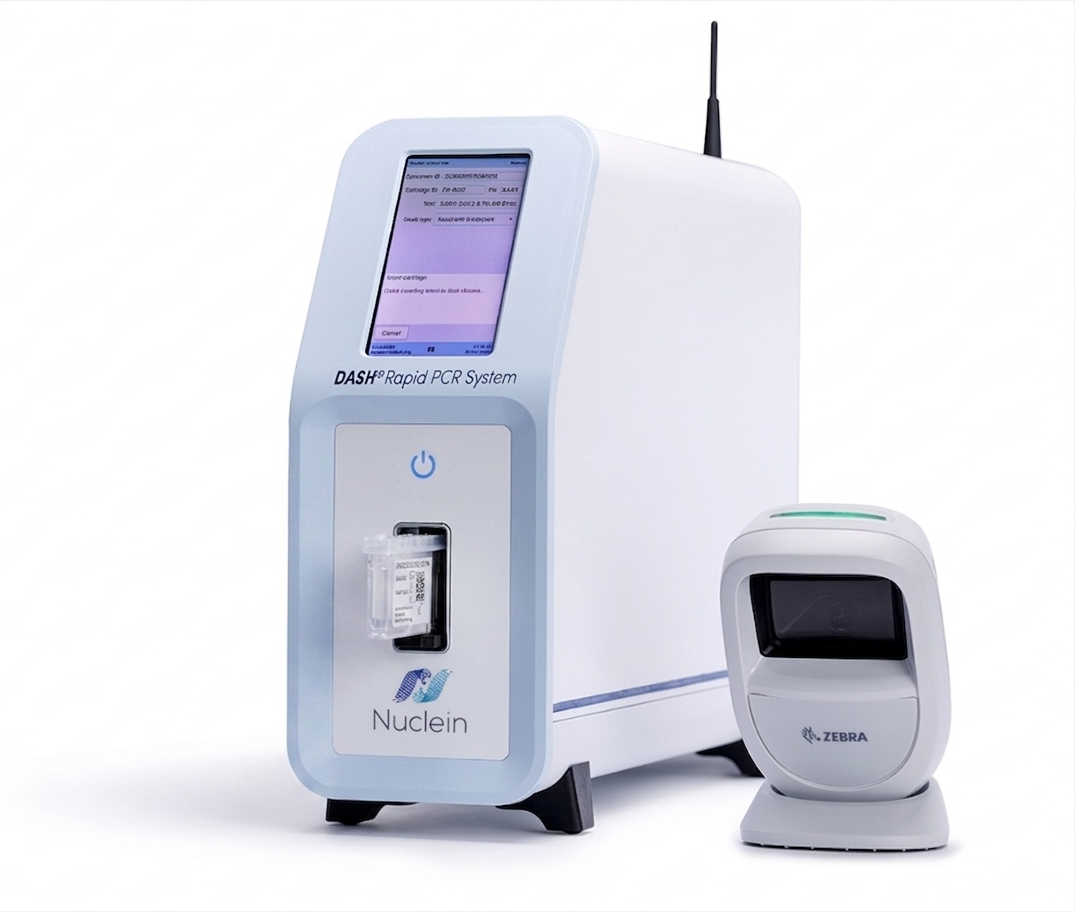
The DASH® Rapid PCR System, is a point-of-care (POC) in vitro diagnostic (IVD) that detects nucleic acids of target pathogens. The first test for the DASH® Rapid PCR System is the FDA-cleared, CLIA-waived DASH® SARS-CoV-2 & Flu A/B Test. The system consists of an instrument, barcode scanner, collection swabs, and test cartridges.
The DASH® Rapid PCR System delivers lab-quality results at the point of care, is simple to use, and can be operated with minimal training.
Assistance provided:
- IEC 62304 Software compliance
- Risk Management Plan & Report
- Preliminary Hazard Analysis
- Hazard Analysis
- Design FMEAs
- FDA Cybersecurity compliance
- Usability Engineering File







.svg)
